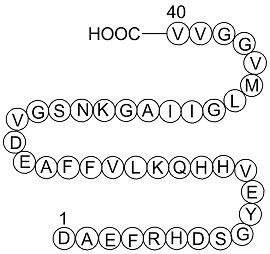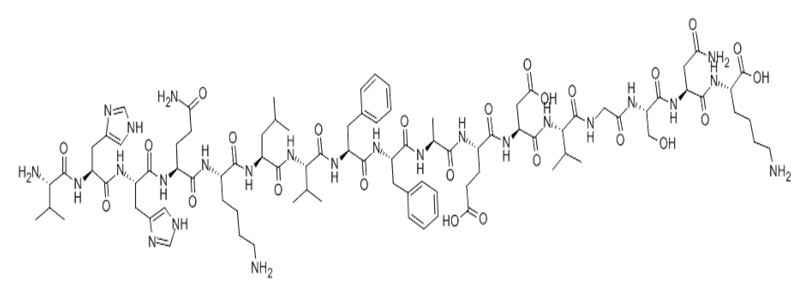
Amyloid Beta-Peptide (12-28) (human) | CAS:107015-83-8 | sequence H2N-VHHQKLVFFAEDVGSNK-OH | High Purity | Manufacturer BioCrick
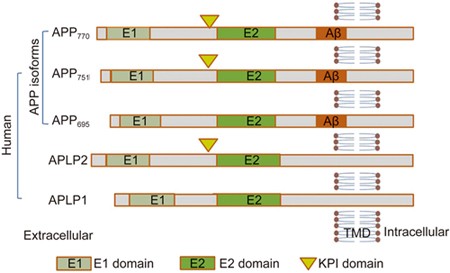
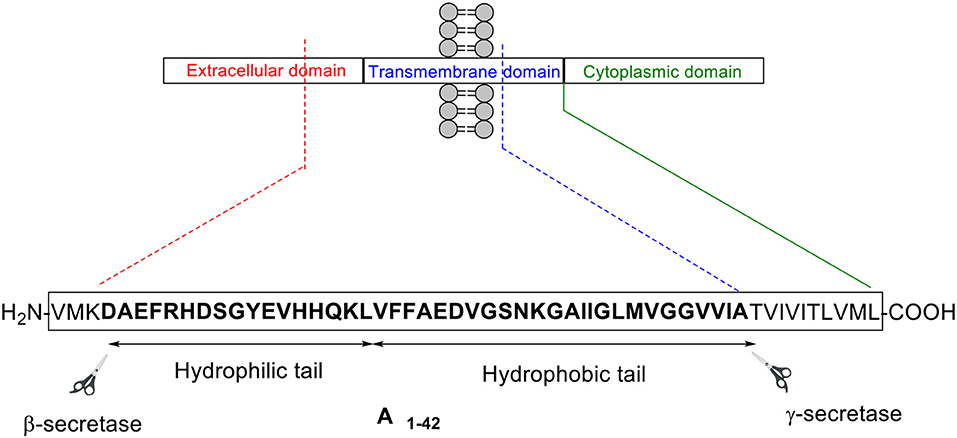
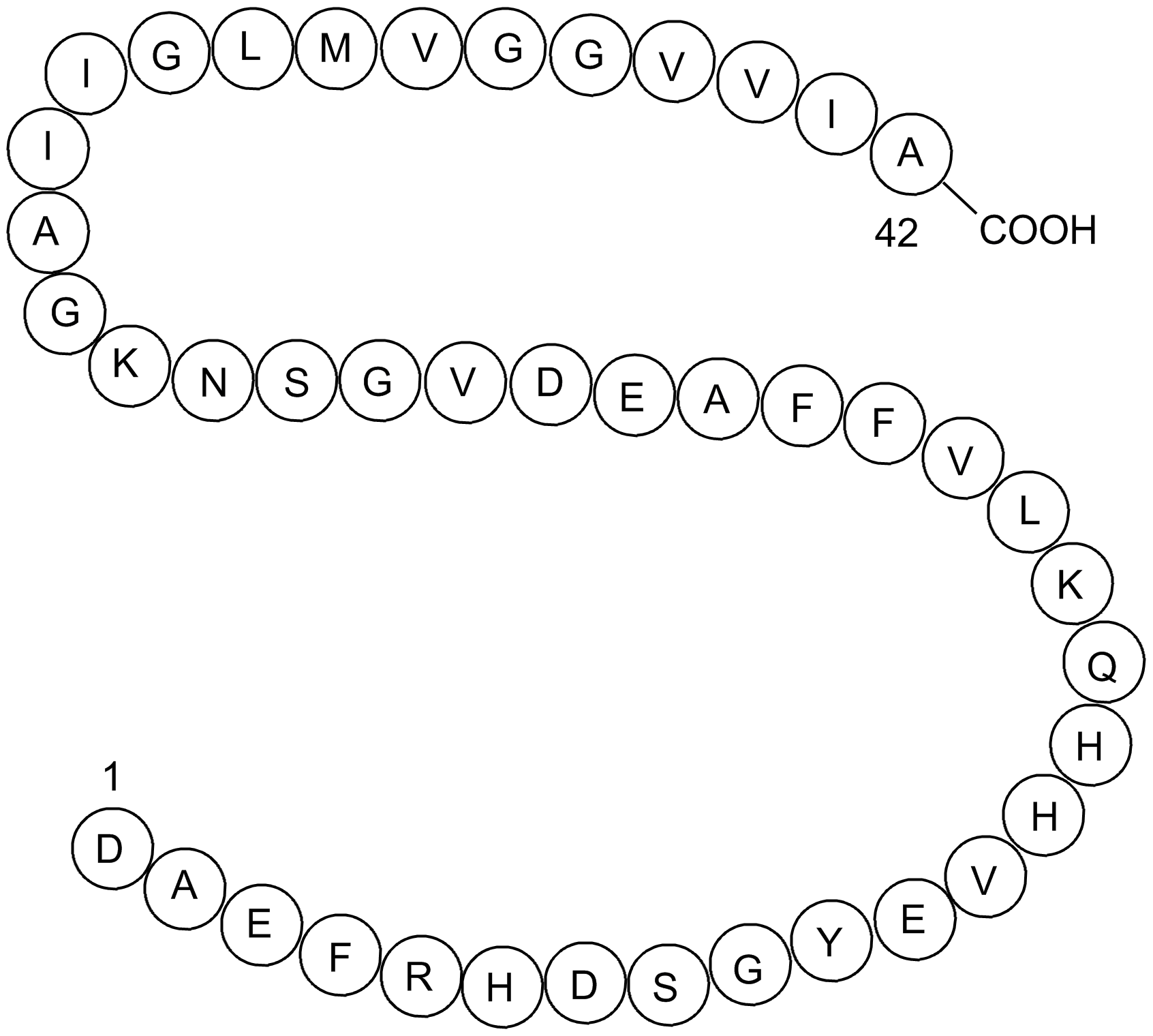
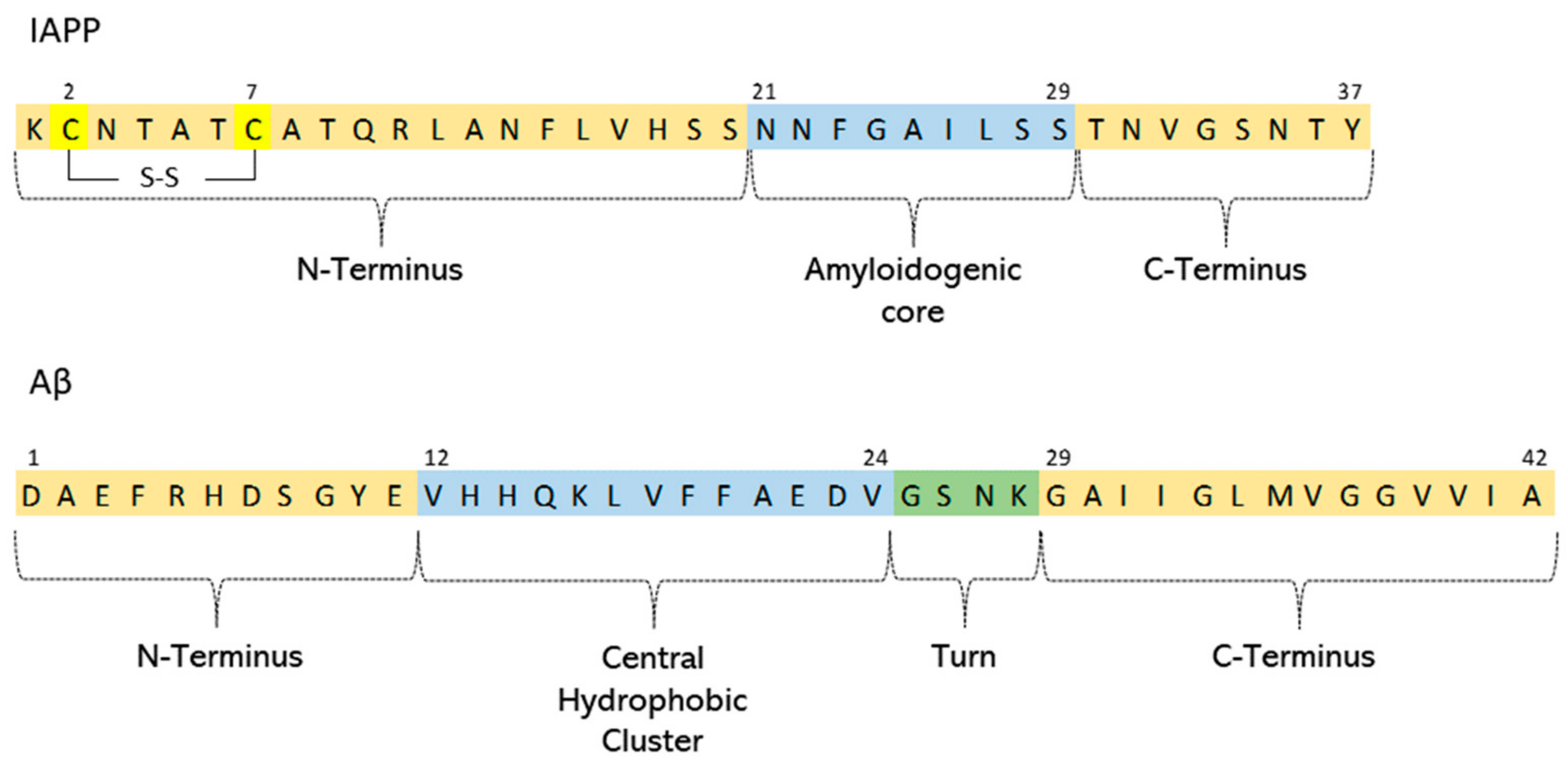
Amyloid beta: structure, biology and structure-based therapeutic development | Acta Pharmacologica Sinica
![PDF] Amyloid beta-peptide (1-42)-induced oxidative stress and neurotoxicity: implications for neurodegeneration in Alzheimer's disease brain. A review. | Semantic Scholar PDF] Amyloid beta-peptide (1-42)-induced oxidative stress and neurotoxicity: implications for neurodegeneration in Alzheimer's disease brain. A review. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e57c7f4364e037d4663e8f59ae915f6a1eb39a9e/2-Figure1-1.png)
PDF] Amyloid beta-peptide (1-42)-induced oxidative stress and neurotoxicity: implications for neurodegeneration in Alzheimer's disease brain. A review. | Semantic Scholar

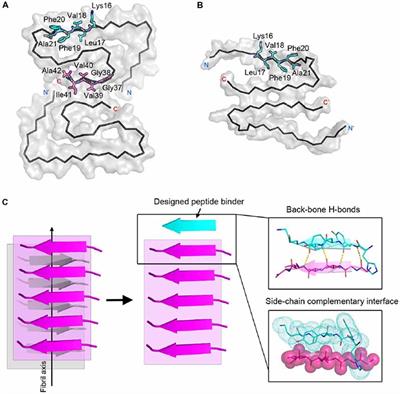
Molecular insights into the surface-catalyzed secondary nucleation of amyloid-β40 (Aβ40) by the peptide fragment Aβ16–22 | Science Advances
Amyloid peptides incorporating a core sequence from the amyloid beta peptide and gamma amino acids: relating bioactivity to self-assembly - Chemical Communications (RSC Publishing)

Mutation-based structural modification and dynamics study of amyloid beta peptide (1–42): An in-silico-based analysis to cognize the mechanism of aggregation - ScienceDirect
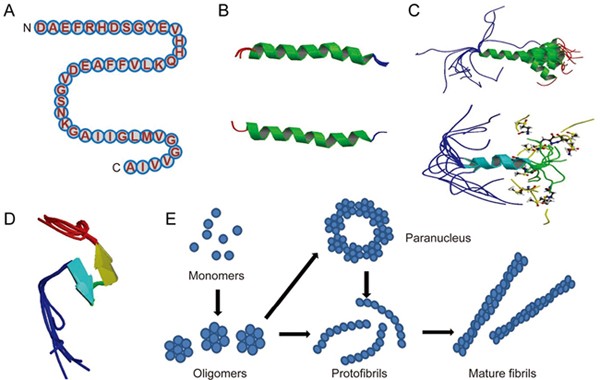
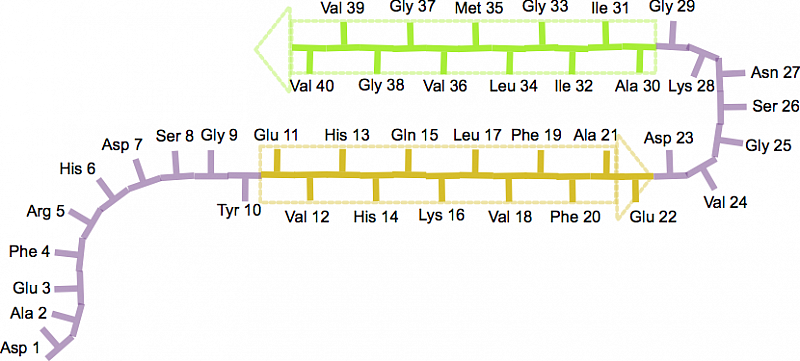
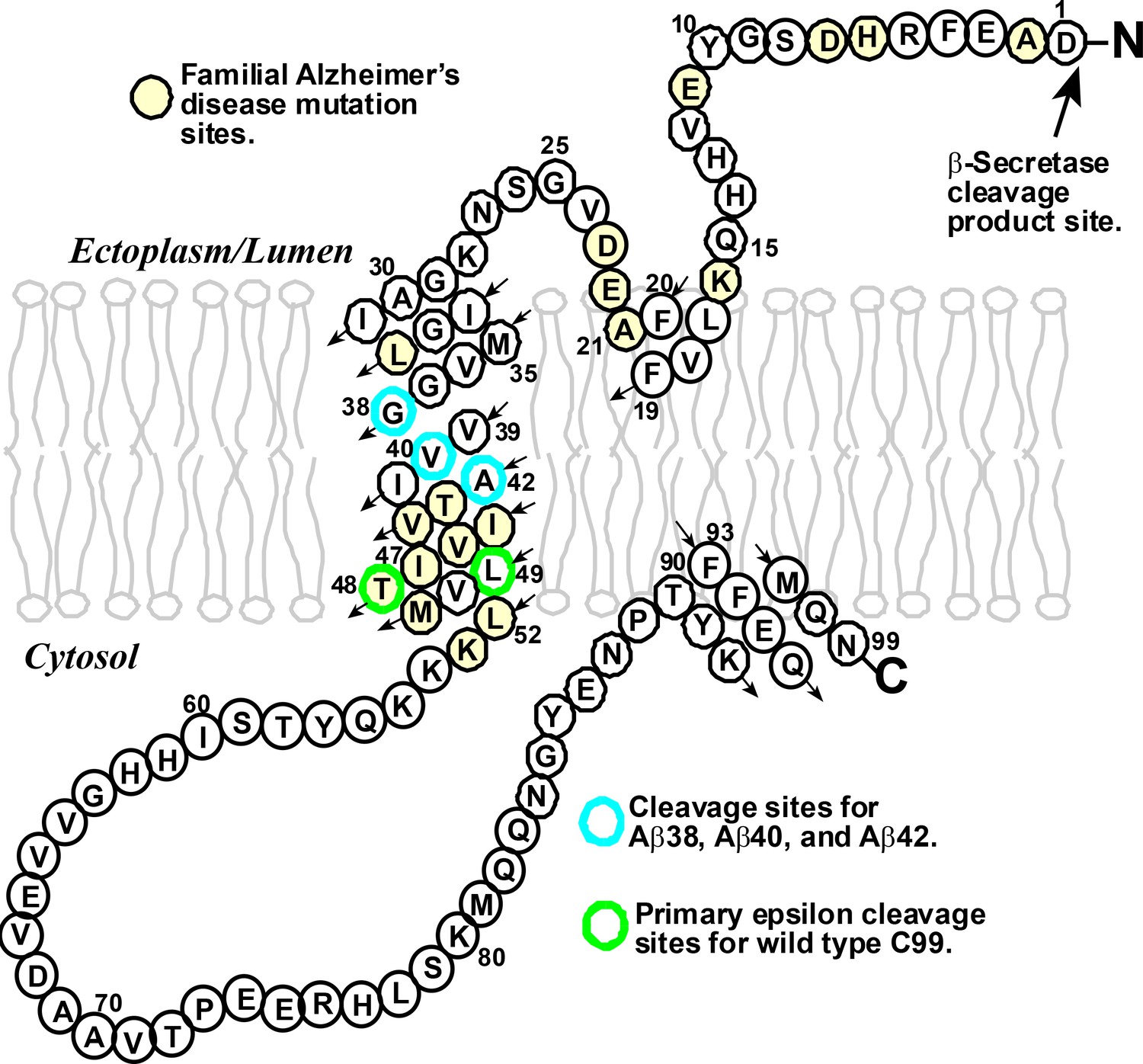
Amyloid beta: structure, biology and structure-based therapeutic development | Acta Pharmacologica Sinica
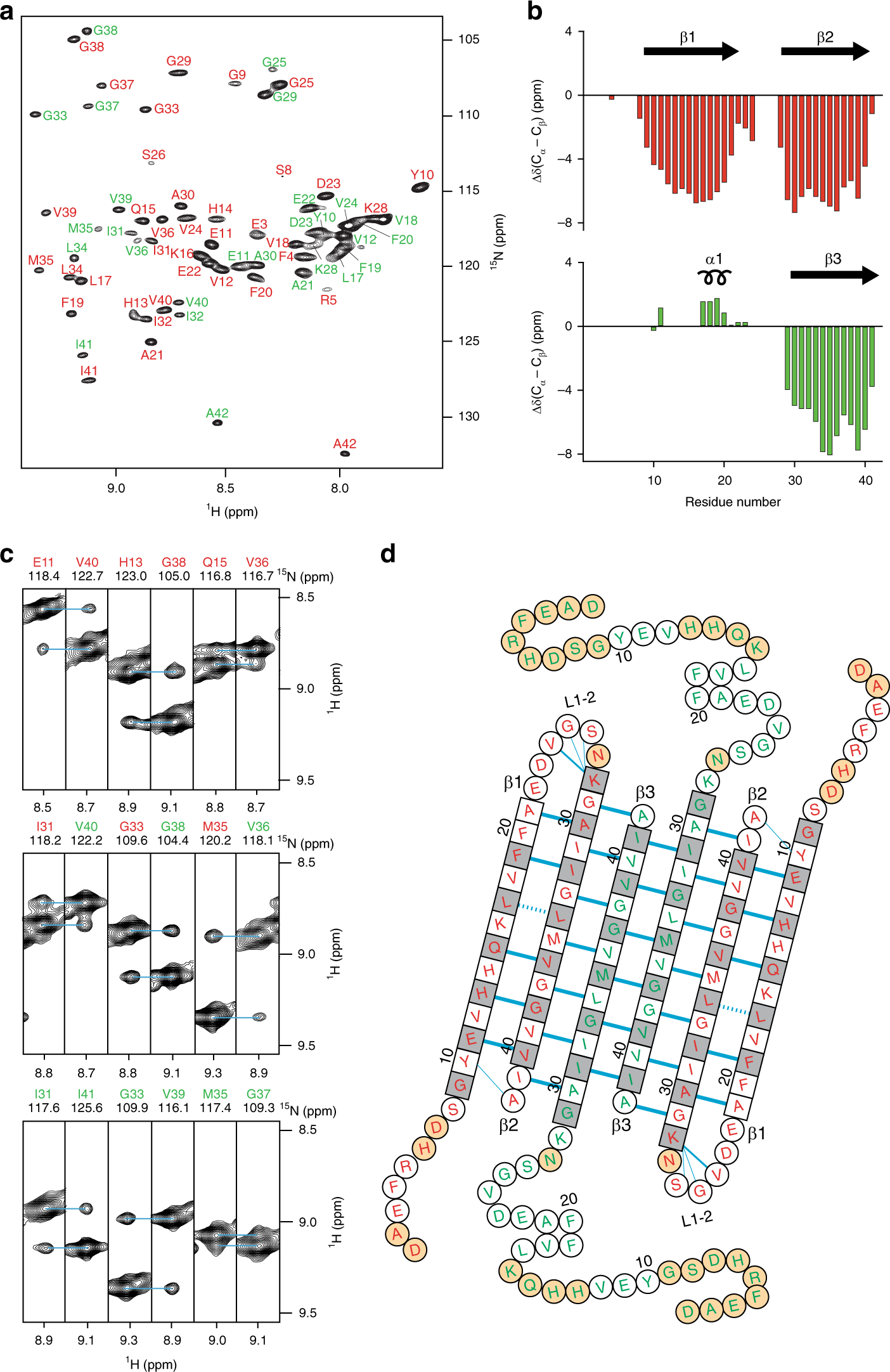
Aβ(1-42) tetramer and octamer structures reveal edge conductivity pores as a mechanism for membrane damage | Nature Communications