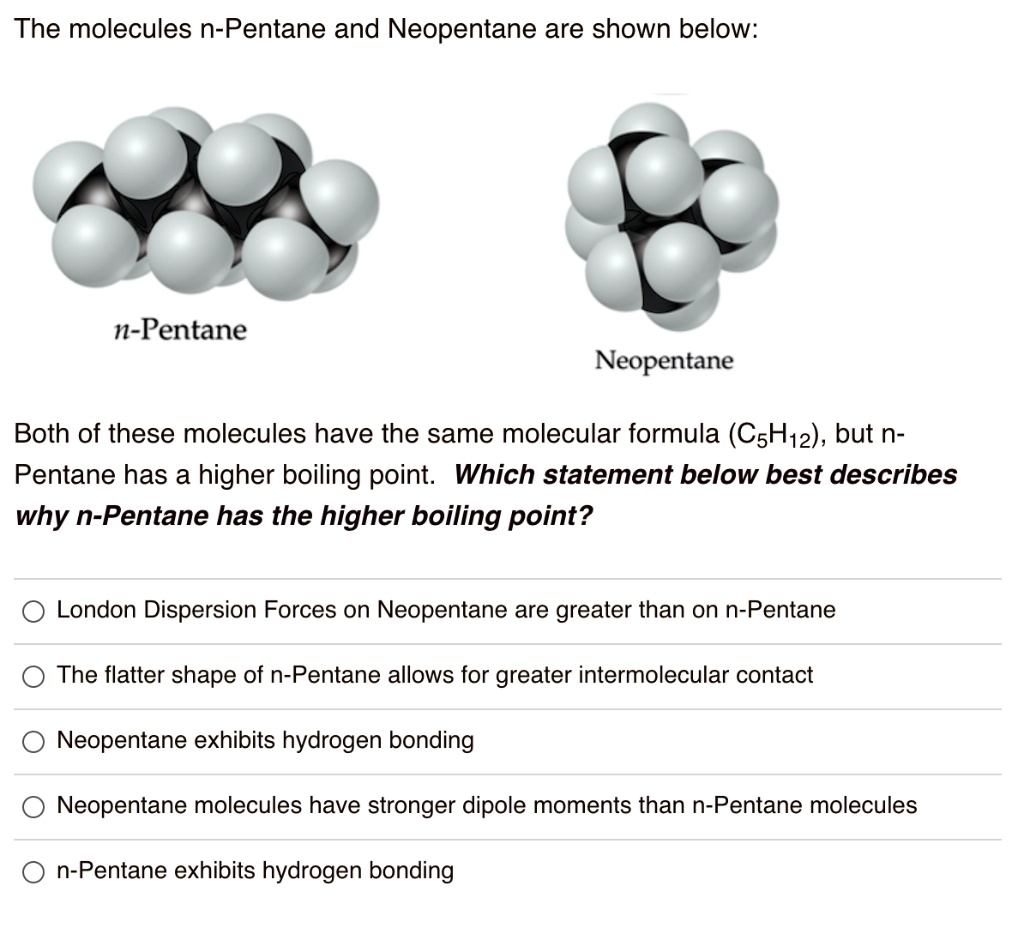
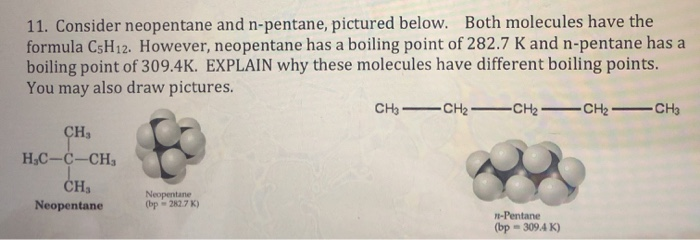
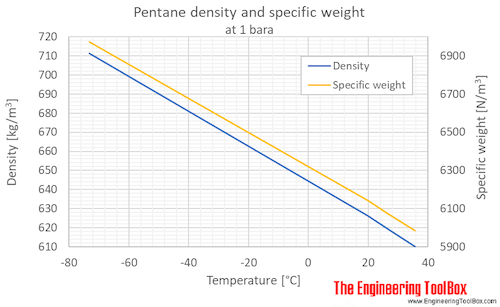
why neopentane has higher melting point than n pentane - Chemistry - Chemical Bonding and Molecular Structure - 13416933 | Meritnation.com
Why Isomers of a compound have different Boiling point (like Isomers of pentane) why force of attraction is not involve in it? - Quora

Pentane has a boiling point of 36.1 degrees Celsius while 1-butanol, which has a similar mass, has a boiling point of 117.7 degrees Celsius. Explain this difference, including line-angle structures of each

SOLVED: 1)The following information is given for n-pentane, C5H12, at 1atm: boiling point = 36.2 °C Hvap(36.2 °C) = 25.8 kJ/mol specific heat liquid = 2.28 J/g°C At a pressure of 1

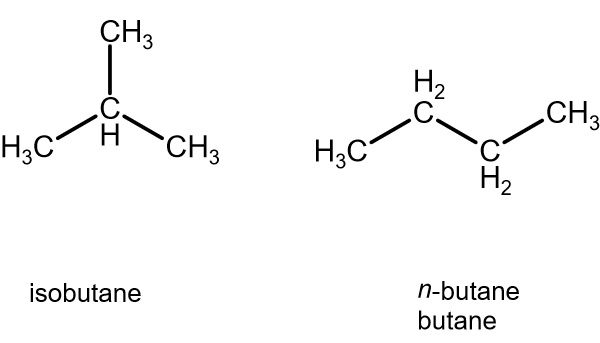
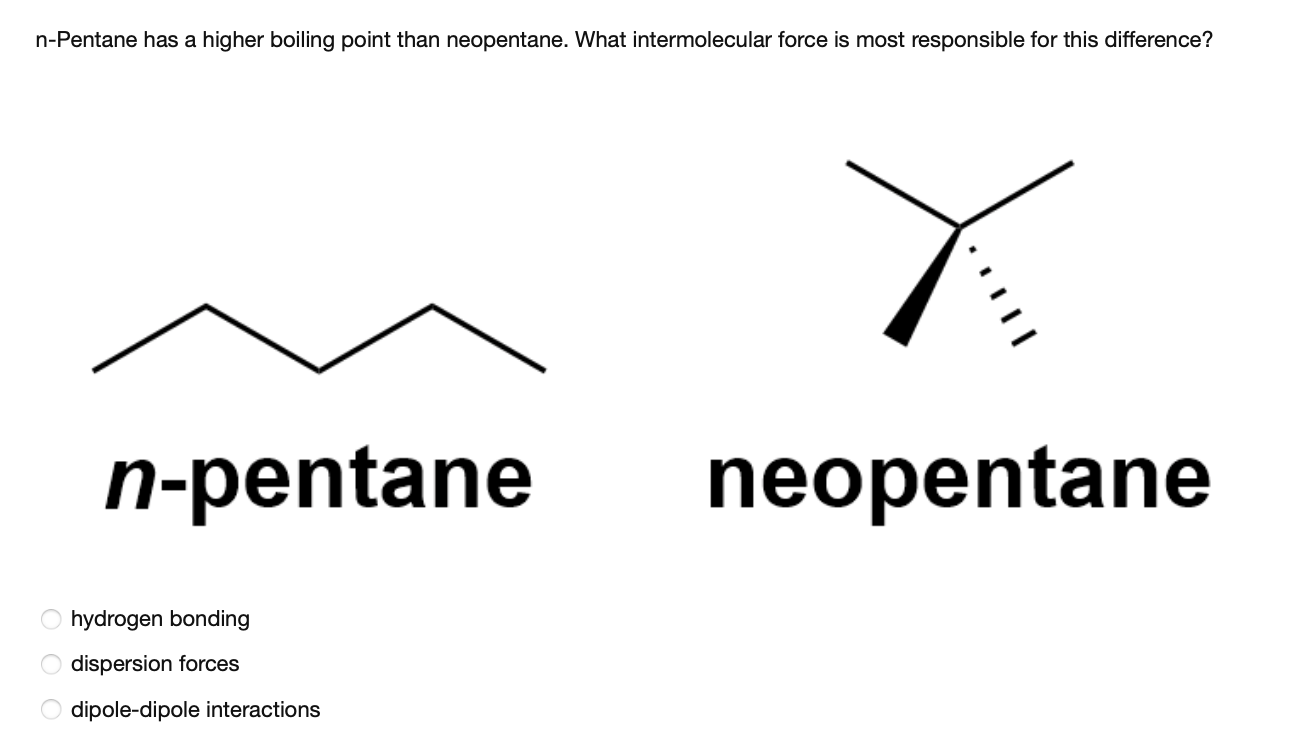
Arrange the following compounds in the descending order of their boiling pointsa) n - pentaneb) isopentanec) neopentane

Rank these compounds from highest to lowest boiling point. a. pentane b. neopentane c. isopentane | Homework.Study.com
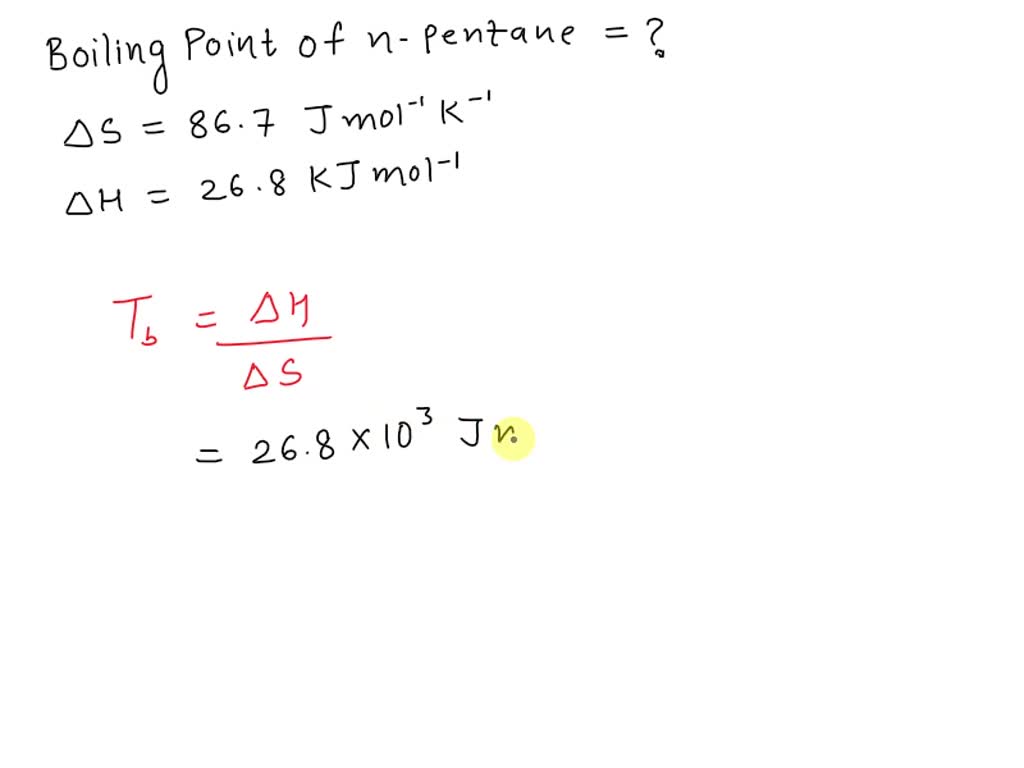
SOLVED: Calculate the boiling point (BP) of n-pentane, given that the = Points) entropy 14)(6= BPis86.7 Jlmol x *K and the change in enthalpy is 26.8 kJlmole change at the

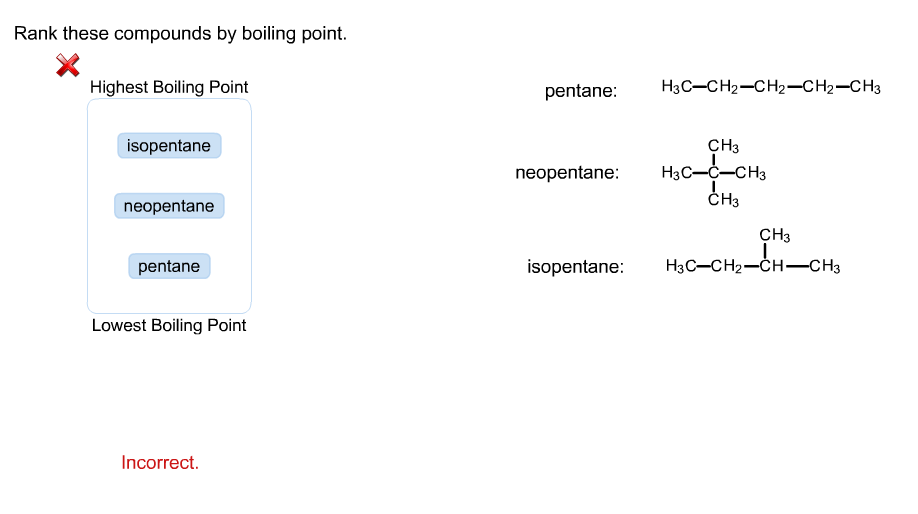
OneClass: rank these compounds by boiling point Rank these compounds by boiling point. Highest Boilin...
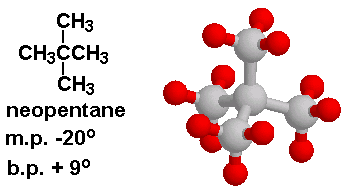
Why is the melting point of neopentane higher than n-pentane but the melting point of isopentane lower than that of n-pentane? Why is the order different than that of their boiling points? -