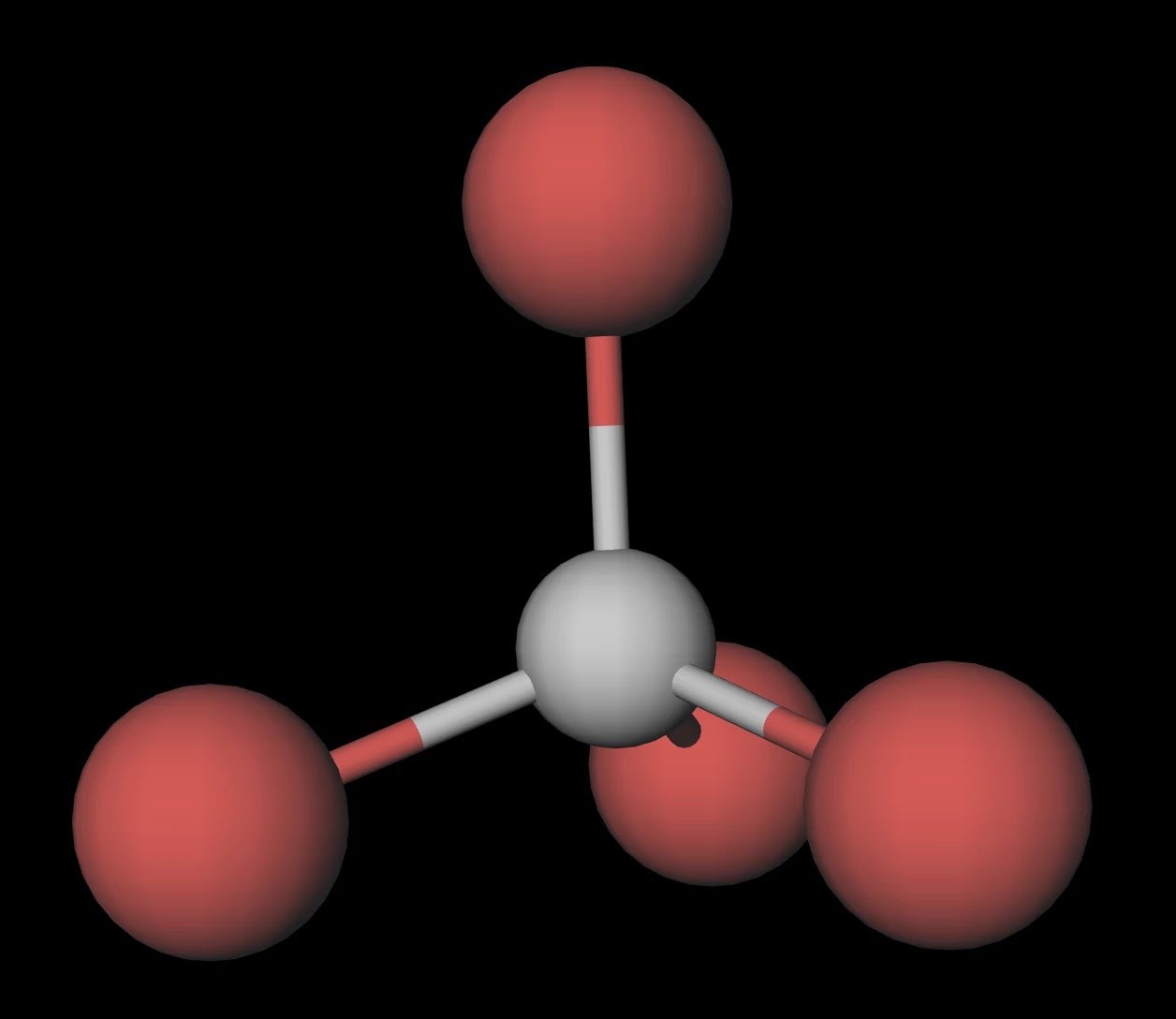
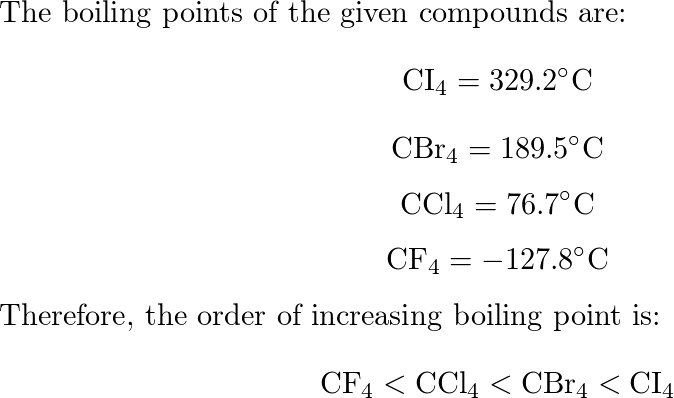SOLVED: C) The boiling point of material is an indicator of the strength of the bonds of the compound: In this case, both CCl4 and CBr4 are covalent molecules with single bonds
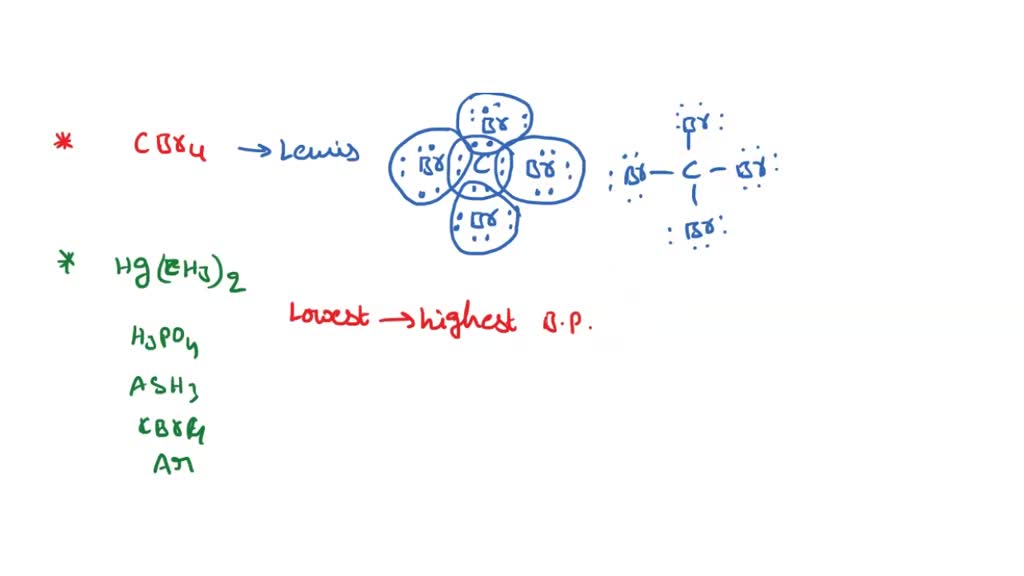
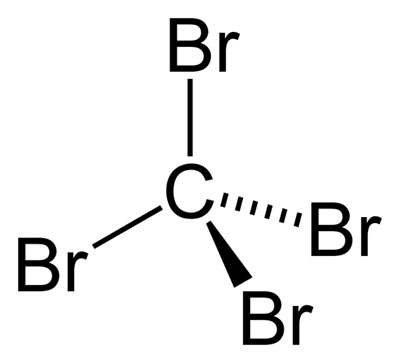
SOLVED: Draw the Lewis structure for carbon tetrabromide, CBr4. Include lone pairs. Arrange the compounds from lowest boiling point to highest boiling point. • Hg(CH3)2 • H3PO4 • AsH3 •CBr4 •Ar

When the substances CCl4, CH4 , and CF4 are arranged in the order of increasing boiling point, what is the correct order?

SOLVED: CBr4 has a zero dipole moment and a boiling point of 189.5⁰C; CH3Br has a dipole moment of 0.05 D and a boiling point of 3.56⁰C. Briefly explain why this polar

SOLVED: In each of the following groups of substances, pick the one that has the given property: highest boiling point: CCl4; CF4, CBr4 b. lowest freezing point: LiF, F2, HCI smallest vapor