What is the order for the boiling point for ortho-chlorobenzene, meta- chlorobenzene and para-chlorobenzene? - Quora

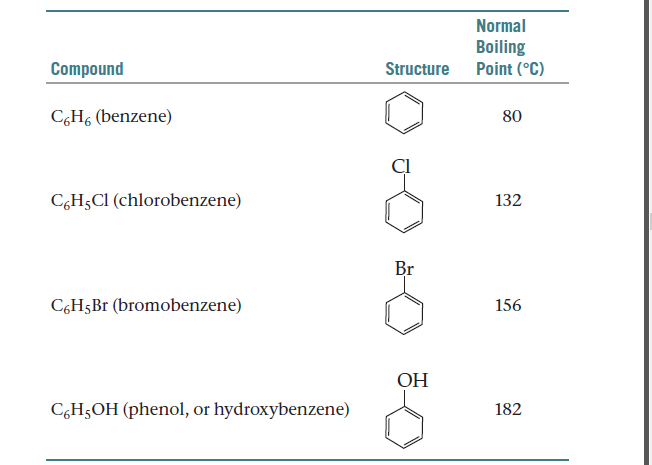
Arrange each set of compounds in the order increasing boiling points. (I) bromobenzene, chlorobenzene, - Brainly.in
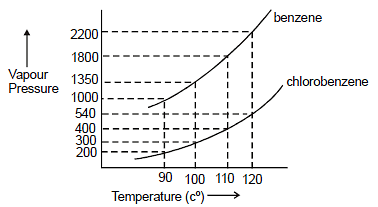
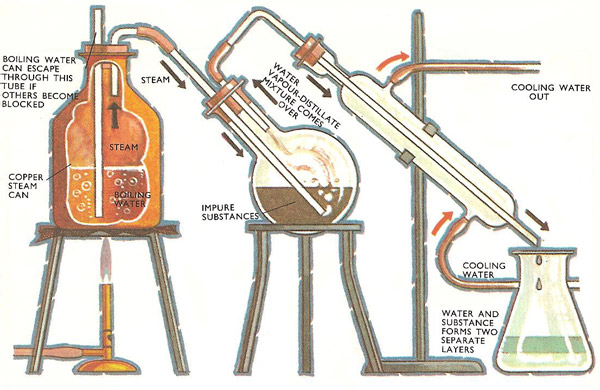
Assuming the formation of an ideal solution, determine the boiling point of a mixture containing 1560 g benzene (molar mass = 78) and 1125 g chlorobenzene (molar mass = 112.5) using the
What is the order for the boiling point for ortho-chlorobenzene, meta- chlorobenzene and para-chlorobenzene? - Quora
Using the following information determine the boiling point of a mixture contains $1560$ gm benzene and $1125\; gm$ chlorobenzene, when the external pressure is $1000\; torr$ Assume the solution is ideal.Given :
1. A mixture is chlorobenzene and water (which are virtually immiscible) boils at 90.3^° C at an external pressure of 740.2mm. The vapour pressure of pure water at 40.3^° C if 530.1mm.

Assuming the formation of an ideal solution, the boiling point of a mixture containing 1560 g benzene (molar mass = 78) and 1125 g chlorobenzene (molar' mass = 112.5)' using the given
1. conversion: aniline to chlorobenzene 2. p dichlorobenzene has higher m.p. then those of o and m isomers. Discuss.