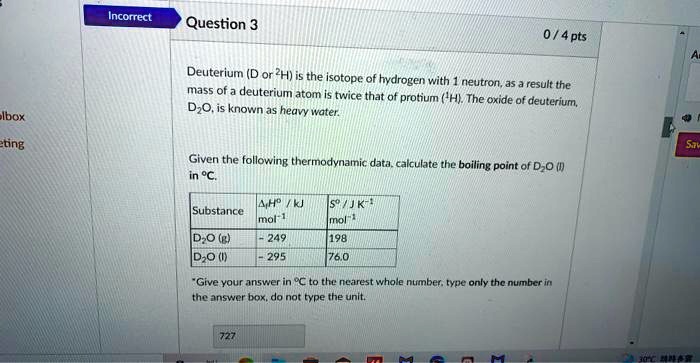
File:Boiling-point-composition diagram for dilute aqueous solutions of deuterium oxide (IA jresv17n6p841).pdf - Wikimedia Commons

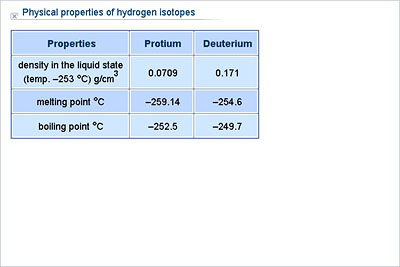
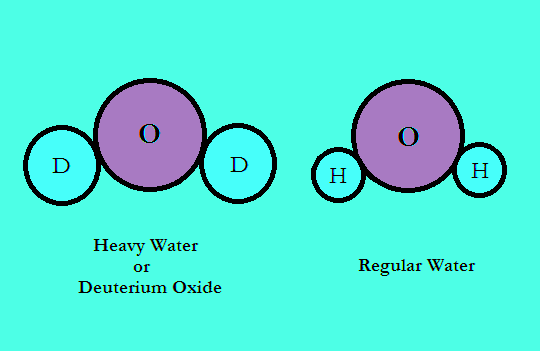
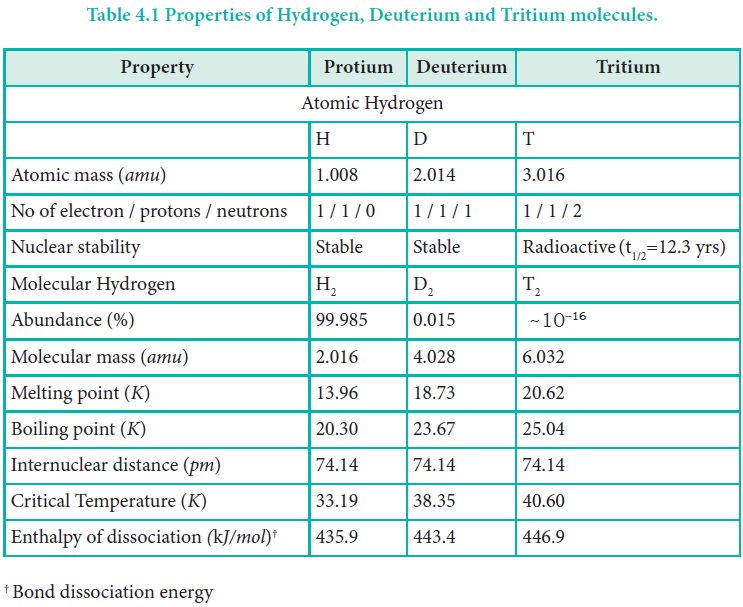
Why is the boiling point of D2O greater than H2O Is it related to hydrogen bonding ! Then please - Chemistry - - 12831129 | Meritnation.com