A split protease-E. coli ClpXP system quantifies protein–protein interactions in Escherichia coli cells | Communications Biology

Expression and purification of a cleavable recombinant fortilin from Escherichia coli for structure activity studies - ScienceDirect

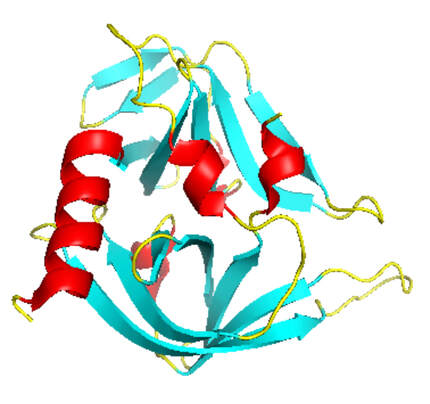
Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE

Directed evolution of the 3C protease from coxsackievirus using a novel fluorescence-assisted intracellular method

Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology
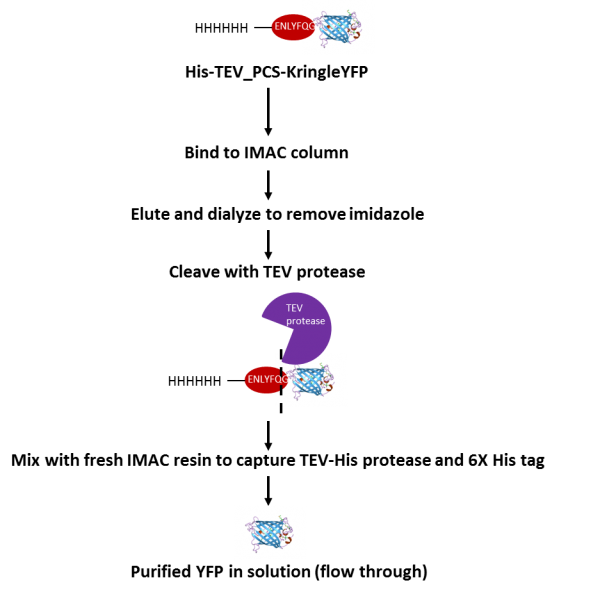
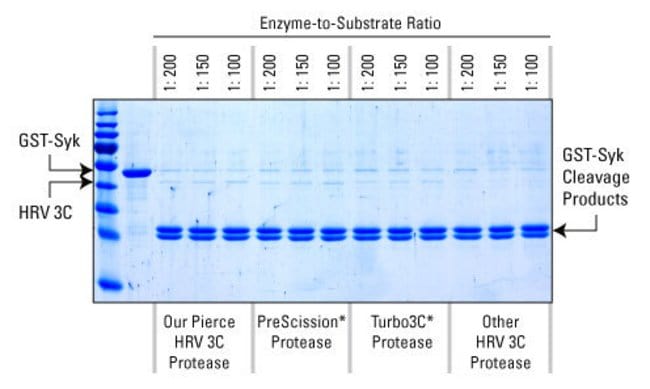
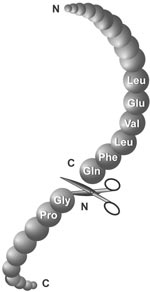
Enzymes - Tobacco Etch Virus (TEV) and Human RhinoVirus (HRV3C) Cysteine Proteases in Vectors | ATUM - ATUM
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE

NT*-HRV3CP: An optimized construct of human rhinovirus 14 3C protease for high-yield expression and fast affinity-tag cleavage - ScienceDirect
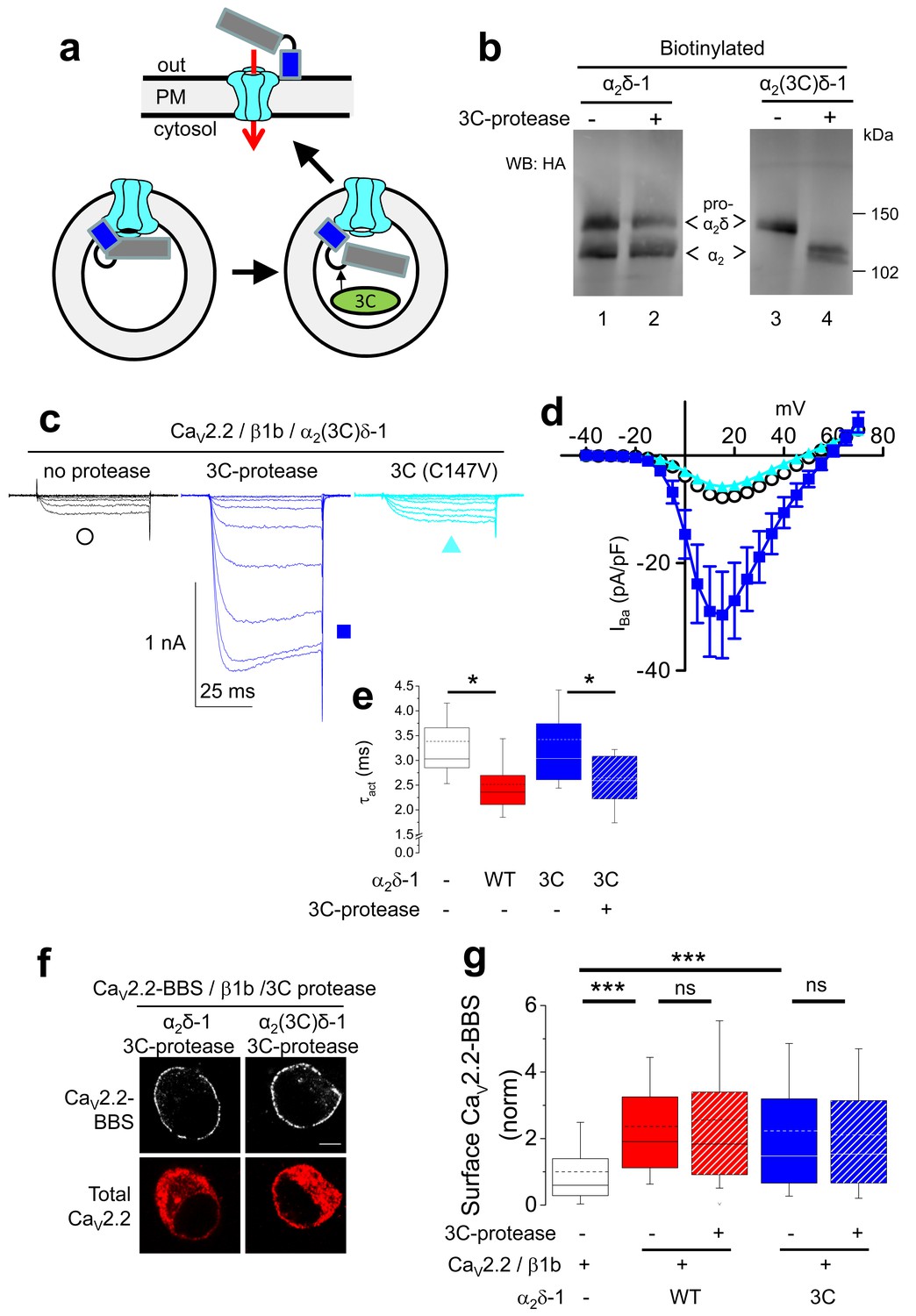
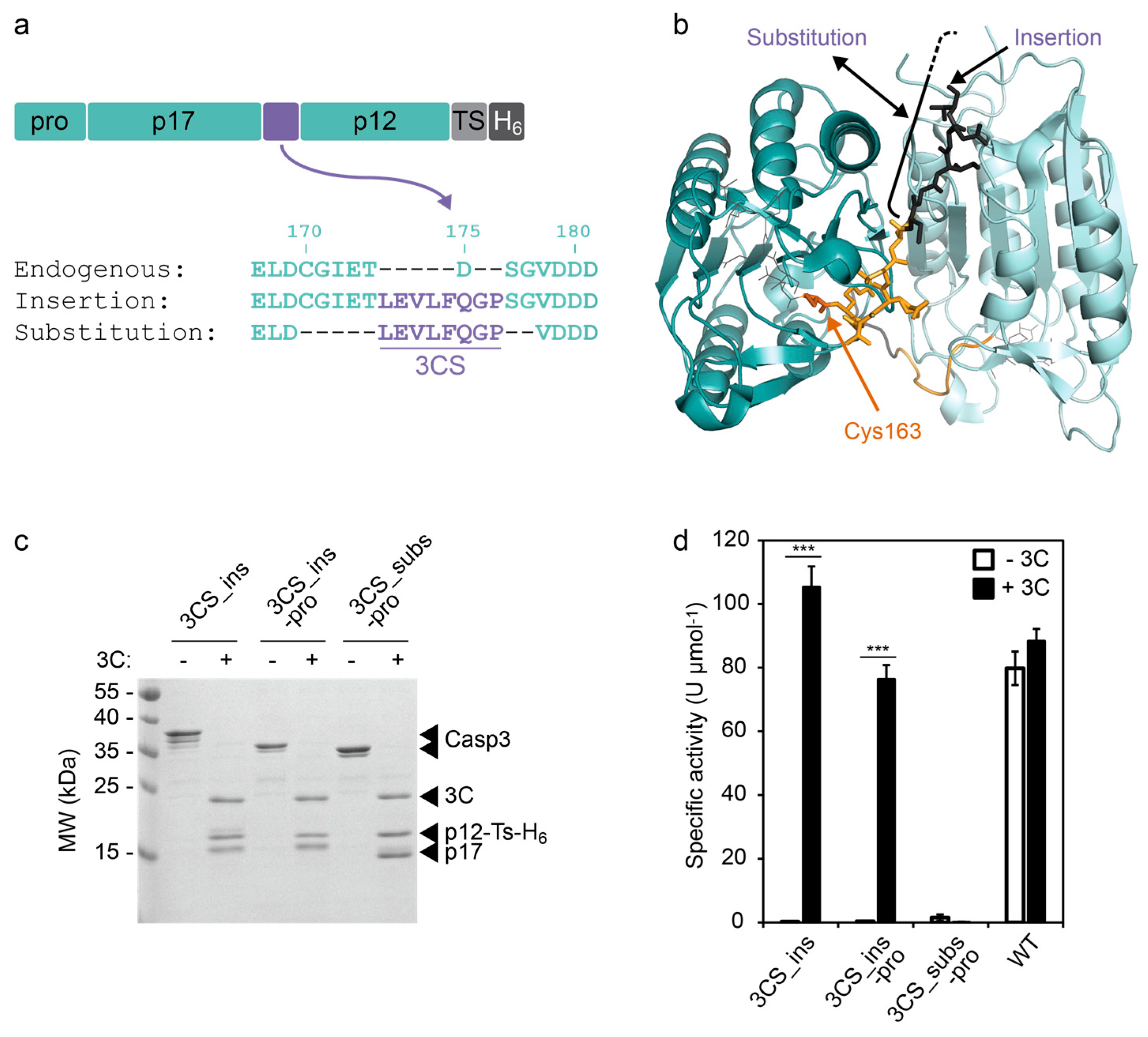
Figures and data in Proteolytic maturation of α2δ represents a checkpoint for activation and neuronal trafficking of latent calcium channels | eLife

Human Rhinovirus 3C protease cleaves RIPK1, concurrent with caspase 8 activation | Scientific Reports