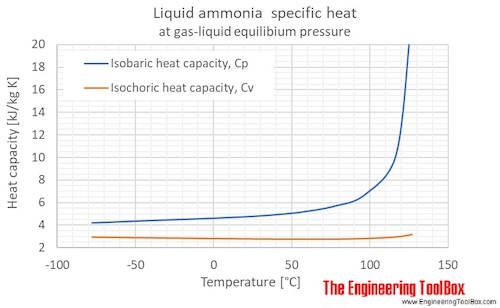
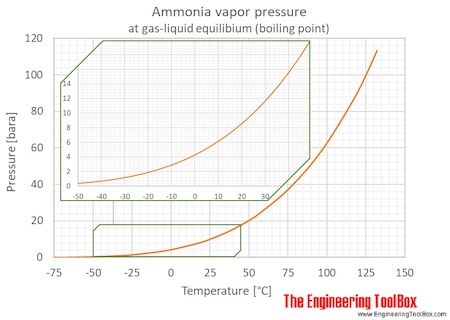
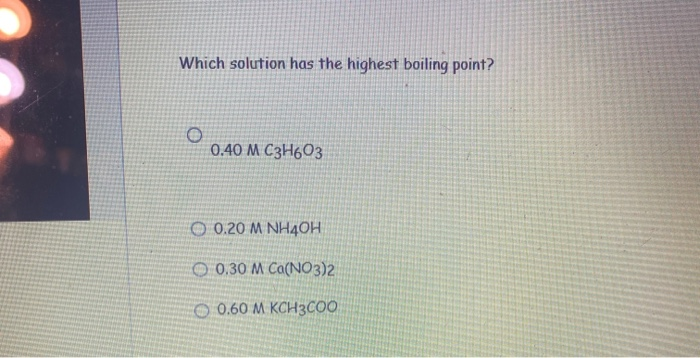
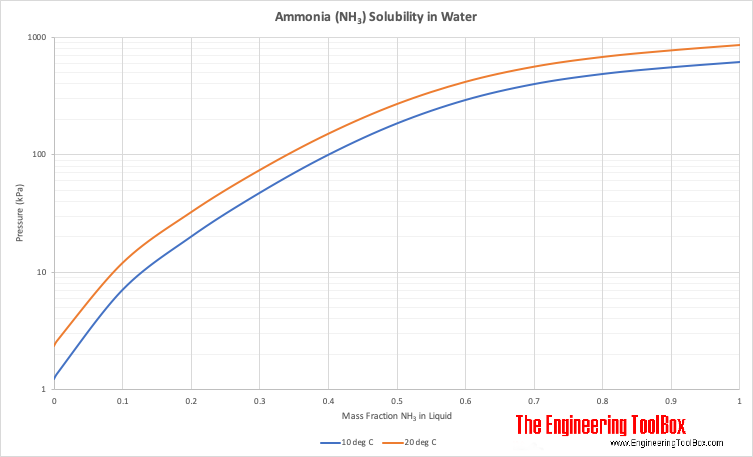
Among the following, the aqueous solution with the highest boiling point is :(A) 1.0M NaOH (B) 1.0M Na2SO4 (C) 1.0M NH4NO3 (D) 1.0M KNO3

home experiment - Does the boiling point of ammonia hydroxide change with the ratio of water to ammonia? - Chemistry Stack Exchange

For 1 M solution of HA, the dissociation constant Ka in terms of vant Hoff factor (i) can be written as :