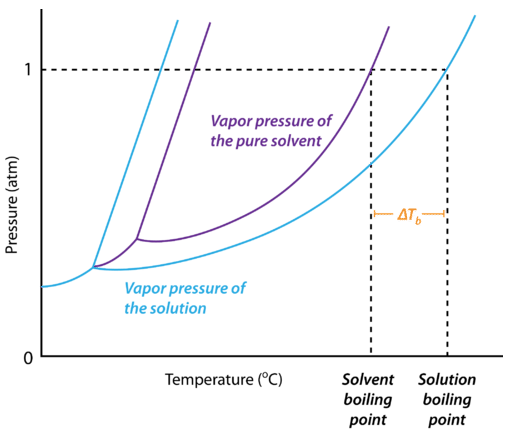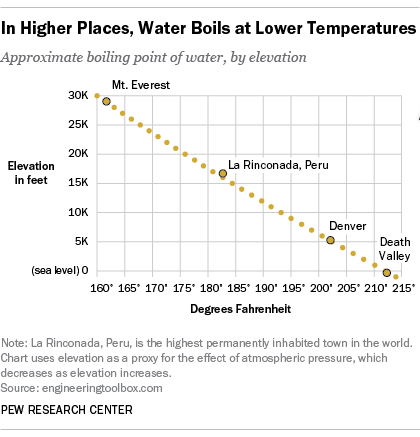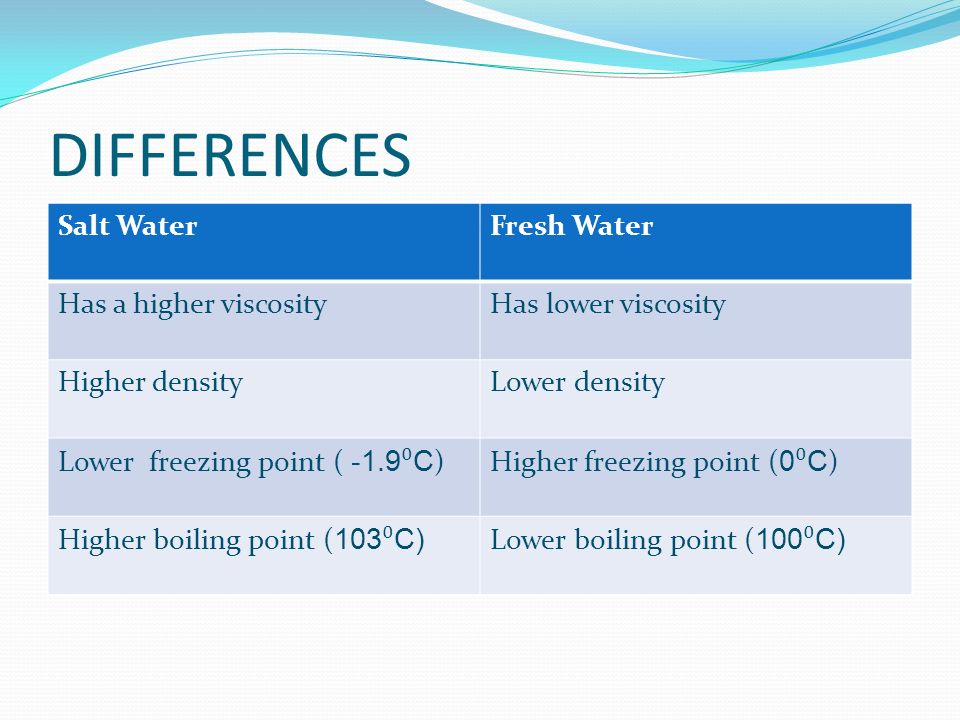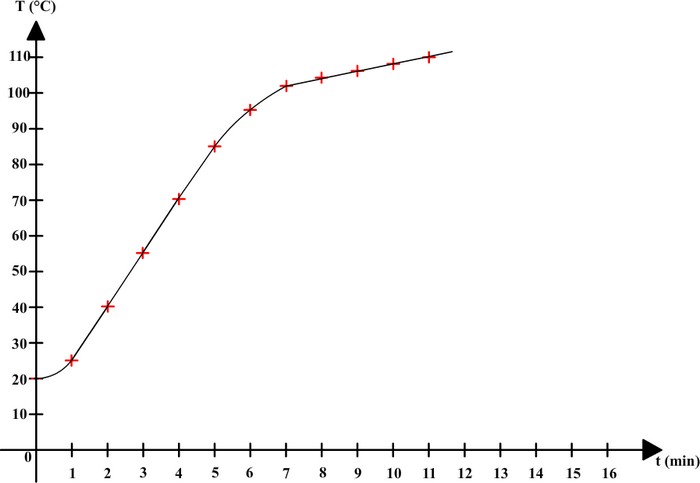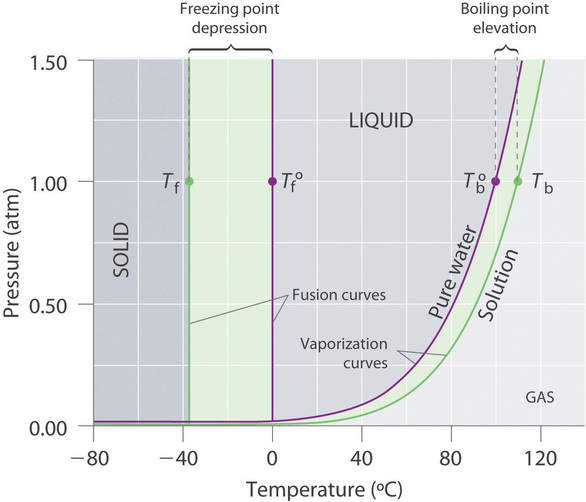
13.8: Freezing-Point Depression and Boiling-Point Elevation of Nonelectrolyte Solutions - Chemistry LibreTexts

Question Video: Identifying Which Properties of a Sample Can Be Used to Help in Testing Its Purity | Nagwa
How does salt increase the boiling point of water? What will happen that makes water molecules less willing to boil? - Quora